Isotopes
Isotopes
Learning objectives
- To identify all isotopes of all known elements.
- To understand the criteria for instability and the different types of radioactive decay.
- To define the decay chains and possible ancestors of a stable element.
- To introduce half-life as a stability criterion
The atomic number Z of an element represents the number of protons present in its nucleus.
- Z=6 for carbon
- Z=8 for oxygen
- Z=20 for calcium
- Z=92 for uranium
The nucleus also contains neutrons. Two atoms can have the same number of protons (same Z), but not the same number of neutrons (N). The mass number A of an element is the number of nucleons present in its nucleus, so A = N + Z. Two atoms having the same Z but not the same A are isotopes of the same chemical element.
Example: The carbon atoms we know are mostly atoms with 6 protons (Z=6) and 6 neutrons (A=12), but there is a very small proportion of carbon with 6 protons (Z=6) and 8 neutrons (A=14). This "carbon 14" is an isotope of carbon.
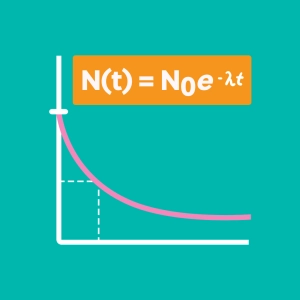
An atom is stable when the number of neutrons is close to the number of protons. If too large a gap occurs, the nucleus becomes unstable and it can undergo one or more radioactive decays in order to regain stability.
The black boxes on the nuclide map in the animation represent stable nuclei. Any atom located outside this zone is unstable and will undergo decays to reach this zone.
Radioactivity is a natural phenomenon in which the nucleus of an unstable atom decays into another, more stable element. During this disintegration, particles and/or highly energetic radiation are emitted. We also speak of ionizing radiation since these radiations have the ability to create ions when they penetrate matter. We know of many applications of these radiations, whether in medicine (nuclear medicine), dating, energy production, the military.
Thanks to the IAEA (International Atomic Energy Agency) for its valuable source of information that is The IAEA's NUCLEUS information resource portal. Its API (Livechart Data Download API) was of great help in developing this educational animation.

Discover EduMedia for free
The interactive encyclopedia that brings science and math to life in the classroom.
Over 1,000 resources