 Electrolysis of water
Electrolysis of water
Learning objectives
- To describe an oxidation-reduction reaction.
- To define the following terms: electrolysis, electrolyte, anion, cation, anode, cathode, electric current
- To know how to read a net equation.
Electrolysis (from the Greek lysis = to split, to dissolve) is the decomposition of a chemical substance under the effect of an electric current.
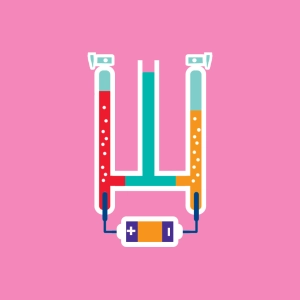
The Hoffmann Voltmeter is an experimental device that enables us to study the electrolysis of water. We speak of the electrolysis of water, but in order to obtain good electrical conductivity, pure water will not suffice, so we have to add an electrolyte, like sulfuric acid (H2SO4. )
Under the action of a potential difference of a few volts, an electrochemical reaction appears at the electrodes:
The cathode releases electrons which decompose the water according to the reduction reaction:
2H2O + 2e- → H2 + 2OH-
At the anode, the water molecules undergo the following oxidation reaction:
2H2O → O2 + 4H++ 4e-
The net outcome gives the following equation for decomposition:
2H2O → O2 + 2H2
The reaction produces twice as much dihydrogen as dioxygen. Avogadro’s Law justifies the fact that the volume of gas in the left tube (H2) is twice as great as the volume of gas collected in the right tube (O2).

Discover EduMedia for free
The interactive encyclopedia that brings science and math to life in the classroom.
Over 1,000 resources





